 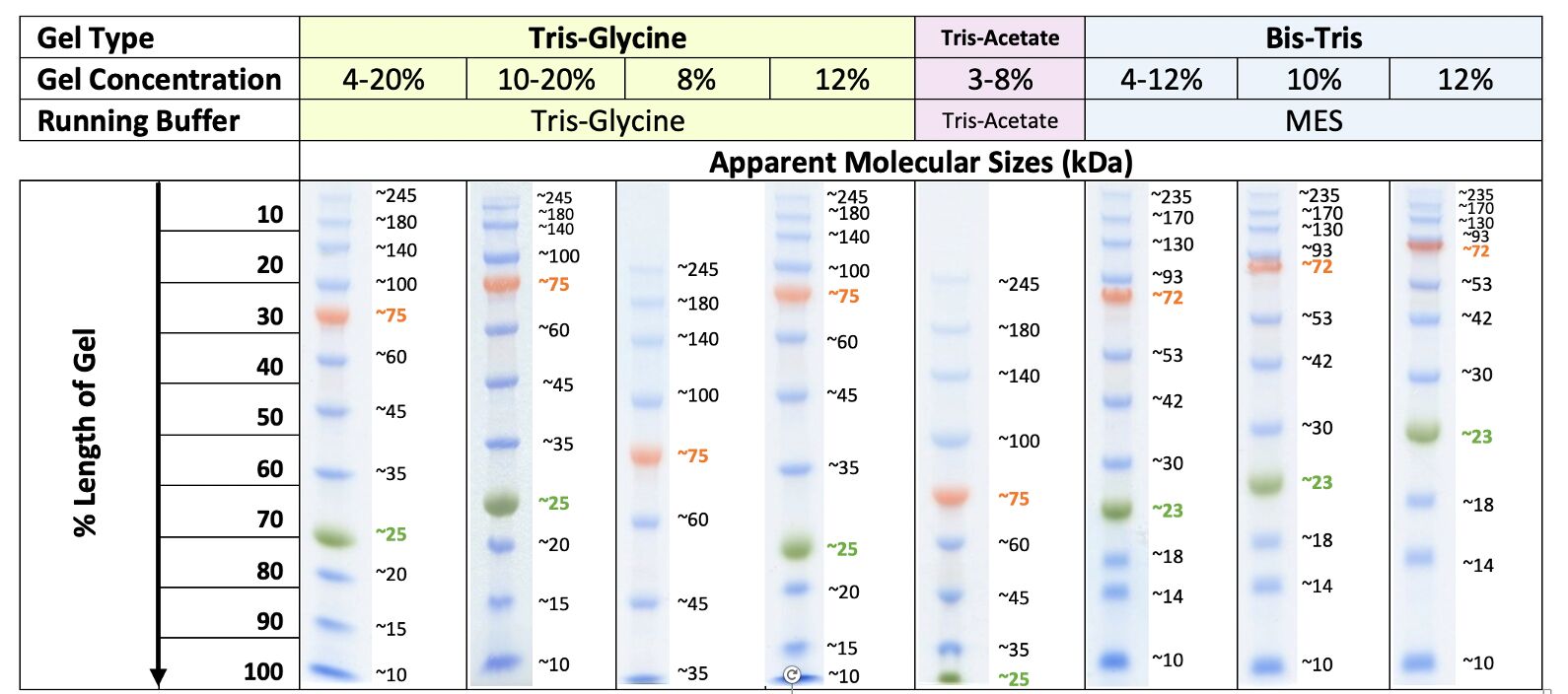
The traditional method to visualize protein ladders on Western blots is to use prestained protein ladders which remain visible when transferred to the blotted membrane. Other home-made protein ladders have tackled the problem of detecting protein ladders on Western blots. Doucet and Beauregard produced a protein ladder by disulfide crosslinking a 11 kD designer protein via oxidation in solution 1. There are a few reported examples of home-made protein ladders. These improvements have come at an increased expense with most commercially available unstained ladders costing about US$ 1.00 per lane. 25, 50 kD) and with optional features such as prestaining with dyes for visibility during electrophoresis and on Western blots. Such ladders have been replaced more recently by recombinant ladders with rounded molecular weights (e.g. 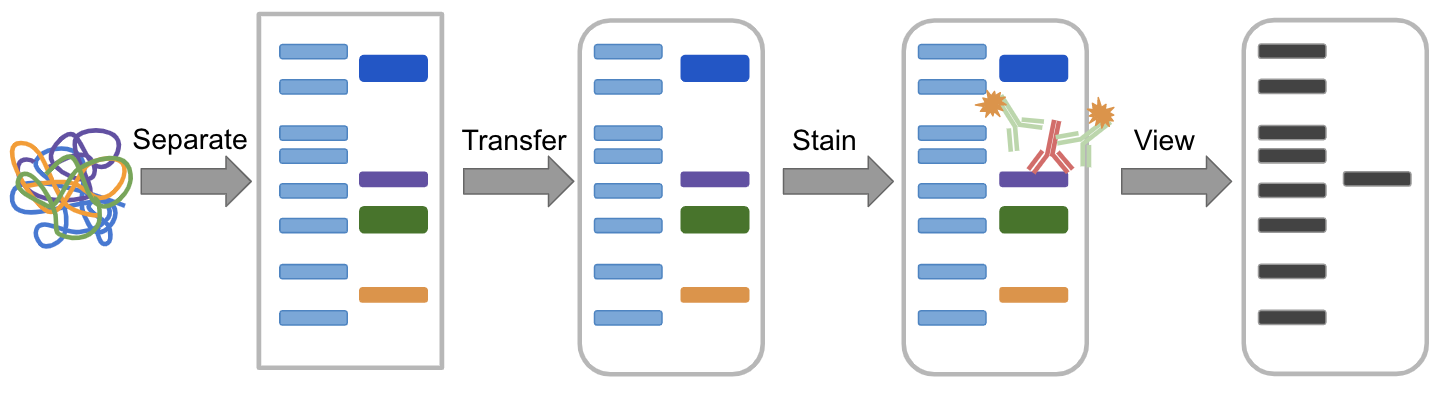
These native protein ladders were commercially available and relatively inexpensive at about US$ 0.10 per lane. As such, protein ladders constitute critical reference reagents when expressing, purifying or analyzing proteins.Įarly protein ladders were comprised of readily available proteins such as lysozyme (14 kD), soybean trypsin inhibitor (21 kD), carbonic anhydrase (31 kD), ovalbumin (45 kD), serum albumin (67 kD) and phosphorylase b (97 kD). They provide molecular weight standards to estimate the size of proteins separated by gel electrophoresis like SDS-PAGE (sodium dodecyl sulfate polyacrylamide gel electrophoresis). Protein ladders or molecular weight markers are among the most commonly used reagents in biochemistry experiments. coli and affinity protein purification, and to research laboratories desiring positive controls for recombinant protein expression and purification. These Penn State Protein Ladder expression plasmids also constitute useful reagents for teaching laboratories to demonstrate recombinant expression in E. 50 ml of culture is sufficient to produce 20,000 lanes of individual ladder protein or 3750 lanes of each set of coexpressed ladder proteins. For more efficient production, we have created two polycistronic expression vectors which coexpress the 10, 30, 50, 100 kD proteins or the 20, 40, 60, 80 kD proteins. We have also constructed plasmids to express 150 and 250 kD proteins. Each protein migrates appropriately on SDS-PAGE gels, is expressed at very high levels (10–50 mg per liter of culture), is easy to purify via histidine tags and can be detected directly on Western blots via engineered immunoglobulin binding domains. The system includes plasmids which express 10, 15, 20, 30, 40, 50, 60, 80 and 100 kD proteins in E. Monitoring protein migration during SDS-polyacrylamide gel electrophoresis.Ģ. Monitoring protein transfer onto membranes after western blotting.ģ. Sizing of proteins on SDS-polyacrylamide gels and western blots.Ħ7 mM Tris-H 3PO 4, pH7.5, 5 mM EDTA, 2% SDS, 33% Glycerol, 0.02% proclin300.We have created the Penn State Protein Ladder system to produce protein molecular weight markers easily and inexpensively (less than a penny a lane). The ladder contains one orange reference band at 70 kDa and a mixture of blue and yellow ribbons at 10 kDa.ġ. The protein marker is supplied in a ready-to-use format for direct loading onto gels no need to heat, reduce, or add sample buffer prior to use.This prestained protein marker is designed for monitoring the progress of SDS-polyacrylamide gel electrophoresis, for assessing transfer efficiency onto PVDF, nylon and nitrocellulose membranes, and for estimating the approximate size of separated proteins that have been made visible with gel stains or Western blot detection reagents. Prestained Protein Marker is a mixture of ten blue, orange or green-stained proteins (10 to 180 kDa) for use as size standards in protein electrophoresis (SDS-PAGE) and western blotting. Membrane-compatible: colored bands transfer to membranes for Western blotting.Two reference bands: orange at 70 kDa and a mixture of blue and yellow ribbons at 10 kDa. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
